YEEDO News
-
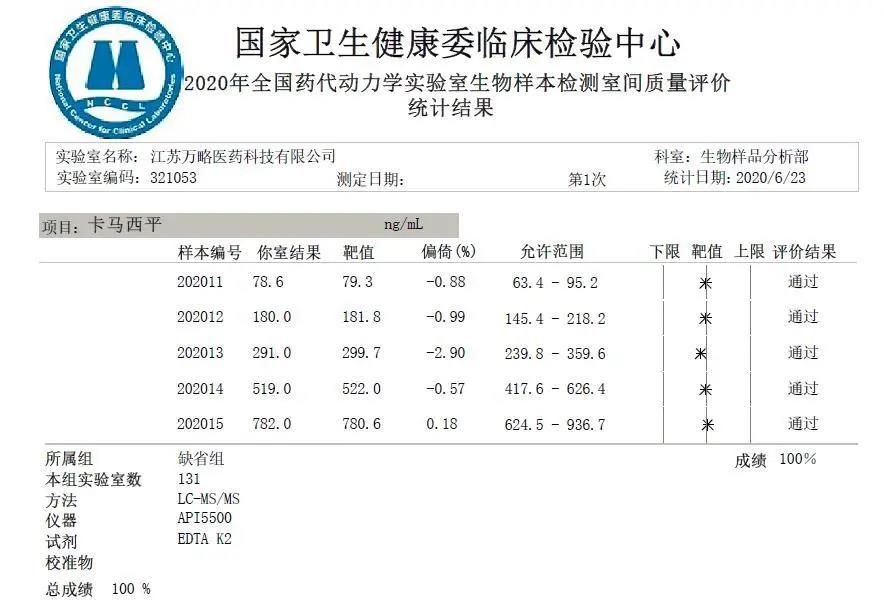
News Express – Jiangsu Value Pharmaceutical Services Co., LTD.
Jiangsu Value Pharmaceutical Services Co., LTD. has won full scores in the external quality assessment by National Center for Clinical Laboratories (NCCL, China) for four consecutive years.2020-08-25 -
First-in-class! China's first original stem cell new drug IND with Yeedozencom registration service!
According to the information released by the Center for Drug Evaluation of CFDA, the autologous reinfusion preparation of REGEND001 cells under Jiangxi Xianhe Medical Technology Co., Ltd. (hereinafter referred to as Xianhe Medical) was approved by the CFDA on July 15, 2020 for the treatment of early and middle idiopathic pulmonary fibrosis (Approval Letter No.: CXSL1900019). The new stem cell drug developed by Xianhe Medical and its parent company Gemfibroon is the first lung stem cell product in the world to enter the clinic. It uses the unique lung tissue regeneration and repair mechanism to play a therapeutic role and is a First-in-class new drug in a real sense.2020-07-23 -
Good news! Yidu Zhengkang assisted Jiangsu Yahong in obtaining the Phase III International Multi-center Clinical Trial Approval Letter for APL-1702 Project
Recently, Yahong Medical Technology Co., Ltd. ("Yahong Pharmaceutical") announced that its international multicenter phase III clinical study of APL-1702 (Cevira ®), a photodynamic drug-device combination product for non-surgical treatment of cervical precancerous lesions, has been approved and issued by China Food and Drug Administration (hereinafter referred to as"CFDA"). In addition to China, Yaron Pharmaceutical has simultaneously initiated this international registration clinical trial in the United States and European countries such as Germany, Romania, Hungary, Russia, and Ukraine, and the results will be used to support registration applications in China, the United States, the European Union, and other countries. Yahong Pharmaceutical is a global innovative drug development company focused on urogenital anti-tumor and related diseases. Dr. Chengfeng Zhuang, chief operating officer and project leader of APL-1702, said. There is currently a high treatment need for women of childbearing age for therapies that can preserve cervical function. APL-1702 is expected to be the world's first non-surgical broad-spectrum treatment for cervical precancerous lesions, filling the important and unmet medical needs and bringing good news to the majority of female patients. "2020-07-20 -

Earth Day | Protect the Blue Homeland of the Galaxy
2020-04-23 -

Yeedozencom: Ten years of hardships and efforts, remain true to our original aspiration and keep our mission firmly in mind
A leading CRO with in-depth and compassionate attitude in China! Your best partner to get your products into China!2020-04-15 -
.png)
Arbor Day | Make our earth more beautiful
Yeedozencom, a CRO service provider of full lifecycle technical services for pharmaceutical products, with in-depth and compassionate attitude!2020-03-17




